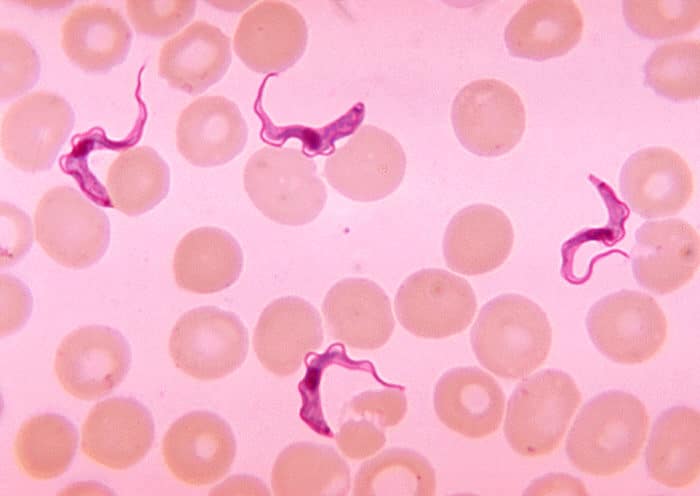
Image Credit: By Photo Credit:Content Providers: CDC/Dr. Myron G. Schultz
Acoziborole, a groundbreaking new treatment for sleeping sickness, has received approval from the European Medicines Agency, marking a significant advancement in the fight against this life-threatening disease. This innovative drug is the first to offer a single-dose treatment, consisting of three pills taken together, and has demonstrated a favorable safety profile in clinical trials, with the only reported side effect being mild to moderate headache.
Sleeping sickness, transmitted through the bite of an infected tsetse fly, affects individuals primarily in remote areas of Africa, particularly in the Democratic Republic of Congo. The disease is caused by the Trypanosoma brucei gambiense parasite and progresses in two stages. The initial phase presents flu-like symptoms, while the second stage can lead to severe neurological damage, including confusion and disrupted sleep-wake cycles. If untreated, the illness can result in coma and death.
Historically, treatments for sleeping sickness have been cumbersome and dangerous, with previous medications causing severe complications for patients. Acoziborole promises to simplify care, as it effectively targets both stages of the disease and can potentially enhance access to treatment in under-resourced regions.
The development of acoziborole was spearheaded by the Drugs for Neglected Diseases Initiative (DNDi) in collaboration with Sanofi, supported by various funding sources, including the Gates Foundation. The drug’s approval follows extensive clinical trials conducted in challenging conditions in the DRC, where health care infrastructure is limited.
In recent years, efforts to control the spread of sleeping sickness have significantly reduced annual cases from hundreds of thousands to approximately 1,000. The World Health Organization aims to eliminate the disease by 2030, and health experts believe that acoziborole will play a crucial role in achieving this goal.
Despite the positive developments, the drug must still undergo review by the DRC Ministry of Health and the WHO before it can be widely implemented. Global health experts emphasize the need for sustained funding and commitment to ensure that communities affected by sleeping sickness receive the necessary treatment and care. The successful integration of acoziborole into existing health systems could represent a transformative shift in the management of this neglected tropical disease.
Check out the original article here: Source link